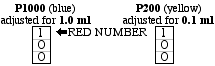Selected Aseptic Transfer ProceduresA guide to the prevention of cross-contamination between environmental organisms and those we really wish to be working on. This page is taken from a handout used in the UW-Madison Bacteriology/Food Science 324 course in the 1990's. Most images are from our old Bacteriology 102 lab manual. |
THE PLATE-TO-TUBE TRANSFER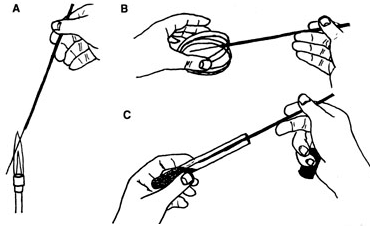
For this and all other transfer situations, the inoculating loop and needle should be flamed slowly from the base to the tip. Note that the petri dish cover is opened just enough to allow entry of the needle. Always when inoculating tubes, the cap or plug is held as shown in the same hand as the loop or needle, and the tube is always held at an angle in the hand – never straight up and down nor standing in the test tube rack. |
THE TUBE-TO-TUBE TRANSFERGo to the narrated movie here. 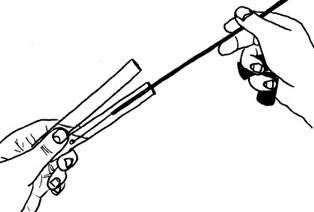
Note that the tubes are held together at an angle. Again, never straight up and down nor standing in the test tube rack. |
THE STREAK-PLATE PROCEDUREGo to the narrated movie here. 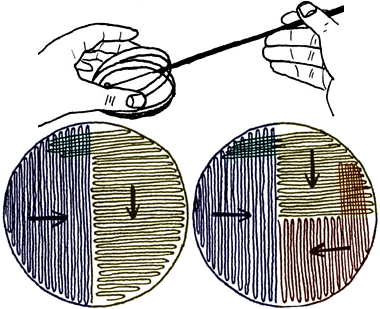
For each "phase," the plate is streaked from the "far edge" (shown) toward the center. Only open the plate just enough to get the loop in, and keep the plate on the table. Further discussion from the Bact. 102 lab manual: The streak plate method is a rapid and simple technique of mechanically diluting a relatively large concentration of microorganisms to a small, scattered population of cells over the surface of the plated medium. The primary purpose is to obtain well-isolated colonies on a large part of the plate surface, each arising hopefully from a colony-forming unit (hopefully consisting of not much more than one cell), such that pure cultures of each desired species in a mixture can be established. Proper streaking of plates is a simple yet indispensable skill required of the bacteriologist. The inoculating loop is the device used for streaking plates. The wire of the loop should not be badly oxidized or pitted or it will fail to "dilute" the inoculum and will scratch the surface of the medium. A fluid suspension, well-shaken to homogenize the cell concentration, is prepared for streaking plates from materials with mixed microbial populations. After flaming the entire wire to redness in a Bunsen burner flame and then allowing the it to cool without touching anything, the loop is tipped into the suspension. If the number of bacteria in the suspension is believed to be small, a loopful is transferred to the plate and the streaking process is begun immediately. If the suspension is believed to have a moderate to dense microbial population, the loop is drained first on the inside of the tube and then applied to a small area of the plate. After depositing this primary inoculum, the loop is flamed and cooled again before starting the streaking pattern. (Whenever desired, a flamed loop may be cooled rapidly by applying it to an uninoculated area of medium at the side of the plate.) Success in obtaining isolated colonies is a function of the amount and density of the inoculum (often very difficult to judge) and the way in which it is streaked. The procedure becomes more of an art than a science, and consistent success is achieved with practice. Two basic patterns are illustrated in the figure above. The three-phase streaking pattern is recommended for the beginning student, because it is most likely to give satisfactory results with suspensions having a wide range of microbial density. To streak the plate, open the top lid just enough to allow the insertion of the loop from the right (from the opposite direction if you are left-handed). See the figure above the streaking patterns. With a wrist motion, move the loop handle back and forth over the surface of the medium, starting from the far left (from the opposite direction if you are left-handed) and moving toward the center of the plate. (Looking directly down at the plate, the loop should appear to be moving in an up-and-down direction.) Use a very gentle and even pressure. The loop should almost retrace its path with each swing as it moves across the medium surface. In the multiphase streaking patterns, the loop is flamed after the first phase is completed, cooled, and (rotating the plate 90° to again streak from the left edge toward the center) recharged by streaking over the first phase a few times. One does not go back to the culture tube for additional inoculum. This is repeated for any subsequent phases. Do not keep the plate uncovered unnecessarily! Replace the lid immediately when you are through streaking the plate, and flame the loop – carefully, as discussed above – before setting it down. |
ASEPTIC TECHNIQUE FOR POURING PLATES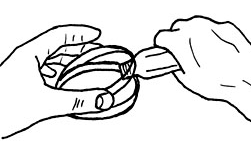
Here we continue the rule to open the plate just enough – in this case to allow entry of the open end of the bottle or tube. |
HOLDING THE PROPIPETTE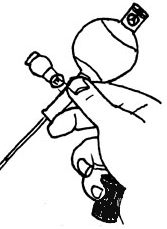
As we generally use the pipettors with tips (below), this illustration applies to us only occasionally. However, the pipettor is held in much the same position: at an angle (as are the tubes). |
HOLDING AND USING THE PIPETTOR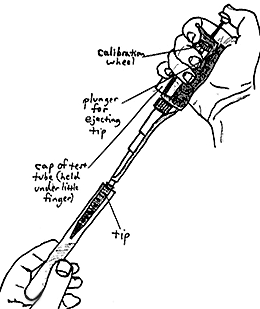
In place of pipettes and propipettes which are often awkward to use and tend to create a lot of waste (i.e., the disposable pipettes), we now use special pipettors which are easily and quickly attached to sterile, disposable tips that produce a relatively small amount of bulk waste. Of the various pipettors which can be utilized, the "Pipetteman" (Gilson) is illustrated here. The P1000 model accepts the blue tips and is usually calibrated for one ml. The P200 model accepts the yellow tips and is usually calibrated for 0.1 ml. Calibration of these amounts is achieved by rotating the wheel shown in the figure, and what should be seen in the window for these amounts is shown as follows:
Note the way in which the pipettor (with the attached tip) is held in the figure. Here, one is about to withdraw material from a test tube or dispense material into it. Also note how the cap of the test tube is held by the hand holding the pipettor. The tip must be replaced before working with another dilution or sample, unless you are moving up to a more concentrated suspension of the same sample. To eject the tip, press down on the plunger (shown near the thumb on the figure), directing the tip to a container of disinfectant. |
| GO TO: |
Selected General Microbiology Topics Archived Bacteriology 102 site Archived Microbiology 102 site Site Outline of our microbiological pages |
These general microbiology pages have copyright by John Lindquist |